Vaccine Success Undercut By Low Power Trial Results
Two potential vaccines for the COVID-19 virus have been announced from Pfizer/BioNTech and Moderna pharmaceuticals with success rates of 95% and 90% respectively. For the Pfizer/BioNTech messenger RNA (mRNA) drug, BNT162b2, it was tested in an equal split, double-blinded trial, and 169 placebo recipients would test positive for COVID-19, whereas only 9 vaccine recipients did.
Pfizer got approval for an emergency use authorization (EUA) from the Food & Drug Administration (FDA) on Friday and a Centers for Disease Control (CDC) panel already recommended it for those 16 and over.
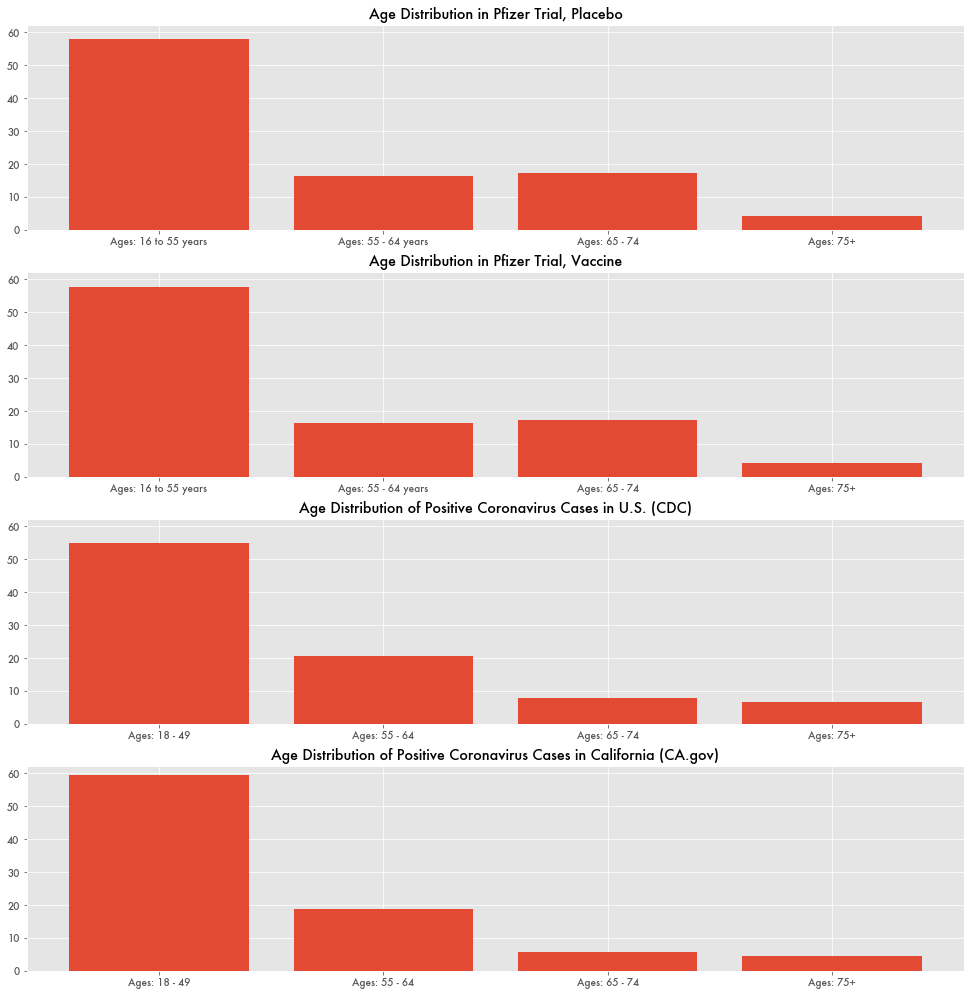
While 169 placebo recipients would test positive, the total population of the placebo group was 40,000, making the infection rate a mere .38 percent, far lower than most reported populations, such as the U.S. general population where the infection rate has been between 6 and 8 percent since June according to numbers from CovidTracking.com. Only 3 of the 40,000 in the placebo group had severe symptoms, which would imply a very low risk of the disease and a low clinical utility to the vaccine.
It wasn't immediately clear why the infection rate was particularly low in the trial. One difference is that the demographics of the trial skewed slightly older than those testing positive in the general population. In both the placebo and vaccine groups, 17.1 percent were between 65 and 75, whereas in the general population it's 7.7 percent.
The Moderna trial had similar results for its mRNA vaccine—a 30,000 person trial with 11 positive tests in vaccine segment and 185 in the placebo group (94 percent efficacy). There were no hospitalizations in the vaccine group yet 30 in the placebo group—putting the hospitalization rate at a surprisingly high 16 percent.
With efficacy rates in the 90s, the results for both are still significant. The FDA has a efficacy threshold for approval of a placebo-controlled trial at 50 percent, with the lowest bound of a confidence interval at 30 percent. A chi-squared test for significance for the placebo versus the vaccine groups returns a p-value far below .01—sufficient enough to say the placebo and vaccine groups are statistically distinct from each other.
But compare those results to the AstraZeneca vaccine trial. Their drug showed only a 70 percent effectiveness, but the infection rate of the placebo population was 1.7 percent—more in line with the infection rate of the general population. And the distribution of tested age groups was also closer to the average age distribution of those who have tested positive for the virus across the U.S.
While the number of vaccine recipients that were ostensibly saved from contracting COVID-19 was only 71—less than the Moderna (174) and Pfizer (160) trial—it was out of a much smaller test population of 5,807. That's around 1/4th the size of the Pfizer trial and implying a more powerful result if the trial was expanded.
Pfizer and Government Funding
Warp Speed is the name for the U.S. government subsidy program to jump start the search for a vaccine. During a recent press conference, Kathrin Jansen, a senior vice president and the head of vaccine research and development at Pfizer mentioned that the company was, unlike their vaccine competition, not part of Warp Speed and “[has] never taken any money from the U.S. government, or from anyone.”
Numerous groups have already pointed out that that statement is not exactly true since the company has received over $590 million in federal tax credits as well as state and local tax benefits, loans and bond financing, and the benefits of using federally financed medical research.
But Pfizer is also known for its offshore tax status, where it uses an inversion—a tax avoidance scheme where one company acquires another company in a low tax country and restructures itself in that country—to keep a large portion of its profits offshore. Pfizer has attempted other mergers, with both Allergan and AstraZeneca, for the tax benefits among other reasons. The Allergan merger was abandoned in 2016 following increased restrictions on inversions by the U.S. Treasury.
As of 2014 it was estimated that Pfizer accumulated $21.2 billion of deferred taxes from this arrangement, yet in 2009 it posted a tax overpayment of almost $770 million.
The company has repatriated large portions of that at different times, such as in 2009 following its merger with Wyeth Pharmaceuticals and in 2005 when there was a tax forgiveness period in place following the America Jobs Creation Act (AJCA). The company's estimated repatriation in 2005 was over $35 billion—the largest repatriation of any company by far, but still only a subset of its international earnings.
In its 2019 financial statement, it approximated its tax liability at $15 billion following the passage of the Tax Cuts and Jobs Act of 2017 (TCJA) that lowered corporate tax rates. The first repatriation installment of $750 million was paid in April of 2019.
And while Pfizer may not receive any direct government grants, it is the recipient of $14.3 billion in U.S. prime government contracts since 2003, for itself and its subsidiaries Wyeth Pharmaceuticals and Cardinal Health. The vast majority of that, over $14.1 billion, was for vaccines destined for the CDC.
For the COVID-19 vaccine, the U.S. is set to purchase 100 million doses for $1.5 billion.